Viruses can be RE-activated by light?
When you’re always looking at old sources, you run the risk of condescending to the experts of the past, who believed scientifically plausible things that now seem obviously wrong. More than once I’ve been ready to point out some amusing practice of the distant past, only to find out that it’s a perfectly valid fact that I (having no medical or physiological training) had never heard of.
One example is “vicarious menstruation”. Is it possible that menstruation could manifest as a nosebleed, or sores in the mouth (sometimes called “herpes”)? Or as a pair of ulcers on the legs, as D.H. Galloway of Roswell, New Mexico reported in 1913? Isn’t it more likely that these stories are exaggerated, or are coincidental? But yes, some combination of hormone levels and blood pressure creates that phenomenon in some women.
Another one is “activated milk”, which contained a substance called “viosterol” that was in high demand for preventing rickets in children. Activated milk? Activated by what?
Ultraviolet light, it turns out. Did this work? Well, UV light turns cholesterol into vitamin D3 when our own bodies are exposed to the sun, and it turns the fungal (yeast) equivalent, ergosterol, into vitamin D2. Cows could be fed UV-activated yeast to make them produce “activated milk”, or activated yeast extract could be directly added to milk. Either of these was a way of “activating” milk that probably worked. Exposure of normal milk to UV light seems like it would be a waste of time.
Whether its benefits were exaggerated or not, activation of milk and other foods was extremely popular, as described in Michael Holick’s great historical review in Public Health Reports, called “The Vitamin D Deficiency Pandemic: a Forgotten Hormone Important for Health”. The drug and food industries fought over whether companies like Fleischmann’s Yeast could claim their products were the equivalent of vitamin D supplements. Here’s a contemporary excerpt from Cartels: Challenge to a Free World, Wendell Berge’s 1944 classic of vaguely paranoid economics.
And all the way into the 21st century, there’s heated debate over whether vitamin D2 (the vitamin D in most supplements) is an appropriate substitute for our own vitamin D3.
* * *
So anyway, here’s another real thing that looked weird and debunkable at first glance.
In virology papers from the fifties and sixties, there are many mentions of something called “photoreactivation”. This started with 1949 work by future Nobel laureate Renato Dulbecco, done in the Indiana University laboratory of future Nobel and National Book Award laureate Salvador Luria. In 1950 Dulbecco summarized the story.
Kelner (1949), working with conidia of Streptomyces griseus, discovered that light belonging to the visible range is capable of reactivating biological material that has been rendered inactive by ultraviolet radiation (UV). Shortly after Kelner’s discovery was known, a similar phenomenon in bacteriophages (bacterial viruses) was observed by accident. Plates of nutrient agar containing UV-inactivated phage and sensitive bacteria had been left for several hours on a table illuminated by a fluorescent lamp. After incubation it was noticed that the number of plaques was higher on these plates than on similar plates incubated in darkness. A short report of this phenomenon of “photoreactivation” (PHTR) has already been published (Dulbecco, 1950).
We’ve been using UV light, gamma rays, and chemical agents like nitrogen mustard to make “killed” versions of viruses, safe for use in vaccines. And now it’s possible that visible light could then re-activate these menaces? Should vaccines be stored in the dark?
Beyond bacteriophages, many other viruses were found to be capable of photoreativation. A sample:
- 1955: “Of the three viruses we studied earlier, tomato bushy stunt and the Rothamsted tobacco necrosis virus showed the phenomenon of photoreactivation, and tobacco mosaic virus did not … Of the six viruses that did [in this study], potato X showed it much the most strongly, tomato bushy stunt and a tobacco necrosis virus the least; cabbage black ringspot, cucumber mosaic and tobacco ringspot were intermediate.”
- 1958: “Thirty minutes of illumination at 300-380 f.c. gave substantial photo-reactivation [of] potato virus X”
- 1961: Tobacco mosaic virus particles can’t be photoreactivated, but RNA preparations from the virus can.
- 1967: “Photoreactivation of UV-irradiated blue-green algal virus LPP-1”
- 1967: “By contrast, photoreactivation of the irradiated [tobacco necrosis virus] was observed in French bean and tobacco, but not in Chenopodium.”
- 1968: Pseudorabies virus can be photoreactivated in chick embryo cultures, but not in rabbit kidney cells.
In the last of those quotes, it’s becoming clear that viruses don’t photoreactivate on their own. They photoreactivate inside cells. You can use UV light to damage the DNA (or RNA) of a virus so it can’t multiply. But it may still infect cells if the protein coat is intact. Then once the viral DNA (or RNA) is inside the cell, the cell’s DNA repair mechanisms can go to work. One of these is photolyase, found in plants, bacteria, fungi, and some animals, but not mammals. Blue light activates this enzyme to reverse the DNA damage caused by UV light (specifically, covalently-linked pyrimidine dimers).
So instead of thinking of photoreactivation as something that happens to certain viruses, we should think of it as something that happens in certain types of cells, to viral DNA as well as cellular DNA.
By 1958, Dr. John Jagger (who does not have a Wikipedia page, though his wife, also a scientist, does) was already able to write a fantastic review of photoreactivation in general (not just viruses and bacteria), saying:
Photoreactivation seems to be possible whether the UV damage occurs in the liquid or the solid state. However, the reactivation seems to require not only the liquid state, but a rather complex environment, similar to that within a living cell.
It doesn’t quite require a living cell, but it requires “cellular material”. A cellular extract still contains the photolyase enzyme.
* * *
You’ll notice that the above examples are almost all plant viruses. This is partially because plants were a very convenient system for virology in the era before cell lines, but it also has to do with the importance of light in plant biology. Dependent on the sun, they need to be able to counteract the negative aspects of ultraviolet light.
But it’s also clear that photoreactivation takes place in insects and fish.
The data show that fish cells have an efficient photoreactivation system at wavelength > 304 nm that reverses cytotoxicity and dimer formation after exposure to filtered sunlamp irradiation of a shorter wavelength (lambda > 290 nm). Shorter wavelengths in UVB (> 304 nm) are more effective in photoreversal than longer ones (> 320 nm). As a consequence, 50-85% of dimers induced by these wavelengths in fish are photoreactivated while they are being formed. A major cytotoxicological lesion is the cyclobutane pyrimidine dimers. Cultured human fibroblasts do not possess such a repair system.
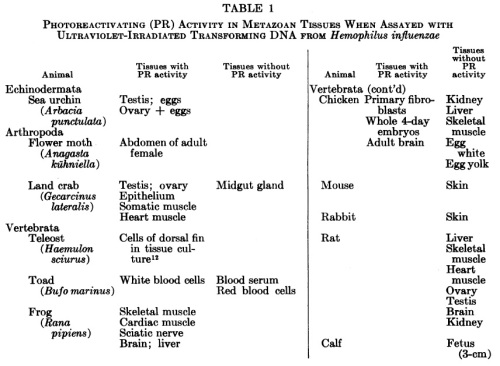
What about that paper above, in which chicken embryo cells enable pseudorabies virus (a herpesvirus) to reactivate? That looks weird, to me at least. Shouldn’t chickens, being warm-blooded animals, be grouped with mammals rather than fish? But chickens aren’t mammals. This table, from Photoreactivating-enzyme activity in metazoa [Cook JS, McGrath JR [1967] PNAS 58(4):1359-1365] sums it up.
Mammals have other DNA repair mechanisms, but we lack photolyase. Which as it turns out, makes us kind of weird.