Sure, put tuberculin in everyone’s eyes.
If you’ve worked in a health care facility, you’ve probably been given the tuberculin skin test. You get a little injection under the top layer of your skin, forming a bubble, and an allergic reaction means you’ve been infected in the past by the tubercle bacillus, or Mycobacterium tuberculosis as we now know it. If you haven’t been infected in the past, you’ll have slight discoloration and maybe slight pain.
Or it may mean you’ve been infected by another species of Mycobacterium. There’s a separate skin test for Mycobacterium avium complex, the “MAC infection” that’s becoming more common, but cases of M. avium often turn up positive from the M. tuberculosis test as well. The material used for the test consists of a purified solution of protein (PPD, or purified protein derivative) extracted from the bacteria.
* * *
The tuberculin skin test is also known as the Mantoux test, and has been for over a century, since Mantoux’s practical application of the hypersensitivity reaction discovered by von Pirquet. There were alternatives for much of that time, all variations on the theme of a small skin injection. The Heaf test, for example, was easier to administer consistently, and probably easier to interpret, but harder to manufacture.
And there were more unusual alternatives, early in the 20th century.
In 1908 three Philadelphia physicians, Samuel McClintock Hamill, Howard C. Carpenter and Thomas A. Cope reported the results of comparisons of several diagnostic tests for tuberculosis. These tests involved administration of tuberculin to different sites in the body: conjuctiva (Calmette); deep muscle (Moro); and skin (von Pirquet).
(from “Orphans as guinea pigs: American children and medical experimenters, 1890-1930” by Susan E. Lederer)
Conjunctiva? That’s… the eye, right? They put tuberculin in the eye, creating an irritation at worst, and a major allergic reaction and possible scar tissue if the test was positive? This was done to people, just as a screening test?
Indeed. Remember, back then a simple injection was not as trivial as it is now. Needles and syringes were not disposable, so the Pirquet test involved scarifying the skin and applying tuberculin into the wound. And if a routine injection led to a hospital-acquired infection, there were no antibiotics to treat it. Dropping some liquid in the eye was easier. More from Lederer’s monograph:
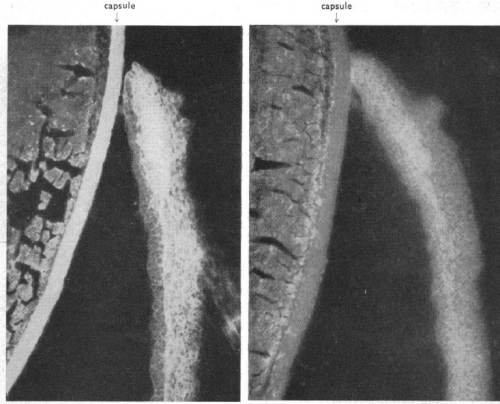
The physicians explained that before beginning the conjunctival test, they were unacquainted with any adverse effects associated with the procedure. The ease of implementing the test (application of a few drops of tuberculin to the surface of the eye) and the relatively quicker results obtained thereby made it attractive to clinicians in search of an effective diagnostic tool. However, in the course of testing, several disadvantages quickly became manifest. The reaction produced a ‘decidedly uncomfortable lesion’ and in several cases, serious inflammations of the eye resulted. In addition, the possibility that permanent impairment of vision might result for several children worried the physicians.
The test proved useful, revealing that many of the children had had undiagnosed cases of tuberculosis. But it was unpopular.
* * *
What were the arguments for and against the eye test?
In the Journal of the Missouri State Medical Association (November 1908), L. M. Warfield explains that the skin test is more sensitive, as it gives positive results from people who have already recovered from tuberculosis, or who show no signs of disease.
This goes along with his instinct for which one is safer: “I have used the cutaneous reaction more than the ocular reaction, for the eye is too delicate an organ to be played with.”
Another complaint about Calmette’s ocular test is that it should not be done on eyes that are suffering any other malady, which is hard to guarantee. In the New England Journal of Medicine (August 27, 1908) Dr. Egbert LeFevre illustrates how complications may arise.
* * *
Within the first year of its introduction the eye test for tuberculosis was already losing fans.
In February 1908, an article by Floyd and Hawes saw the eye test as safer than the skin test — they could be summarized to say “the advantages of the ophthalmo-tuberculin reaction over the cutaneous or subcutaneous methods is that it is absolutely painless, whereas both of the others are painful or disagreeable to say the least. Practically no constitutional symptoms follow the use of the eye, whereas in the subcutaneous test they are important to obtain and often very distressing, and also occasionally occur in the cutaneous method.”
Six months later, doctors were abandoning the procedure. T. Harrison Butler of Coventry, England laid out the empirical observations that changed his mind in the August 8, 1908 British Medical Journal.
Further argument against the eye test came from L. Emmett Holt of New York, whose paper in the January 1909 Archives of Pediatrics (along with the Philadelphia one mentioned above) became a massive controversy when publicized by “anti-vivisection” activists. The title is a bit alarming. (“Babies Hospital” is now called Children’s Hospital of New York-Presbyterian.)
According to Holt, not only does the eye test produce unnecessary discomfort, it’s actually harder to perform.
In ease of application there is a decided advantage in the skin test. The scarification is a trifling thing. The patient does not require continuous observation before or after, and the reaction lasts a considerable time. The ophthalmic cases need closer watching, the reaction is shorter and may be missed. It cannot be used well in ambulatory patients.
The 1909 Eye, Ear, Nose and Throat annual points out yet another practical limitation.
Still optimistic about the eye test, the New York State Journal of Medicine blames problems on improper technique.
In considering the ophthalmic test we must call attention to the fact that harmful results are in all probability due to the instillation of tuberculin into diseased eyes, to infection after instillation, or mechanical irritation, to the introduction of secretion by the fingers of careless patients into the untested eye and to the use of poor or faultily prepared tuberculin.
Calmette reports 13,000 instillations and states that in no case in which the tests were properly applied and controlled were there serious complications. Petit tabulated 2,974 instillations with no ill effects in 698 positive reactions. Smithies and Walker in 450 instillations in 377 patients had four stubborn reactions. It is wise to remind the profession that the eye needs to be thoroughly examined before the test is made and with the slightest abnormality, tuberculin should not be used.
It’s agreed that the test shouldn’t be given to people with any eye problems, and it can’t be given more than once on the same eye (in a lifetime?), and it shouldn’t be given to old people. And maybe you should keep some cocaine around to numb the eyes of children and “sensitive adults” so they don’t squeeze the irritant out of their eyes.
With all these limitations, you’ll have to learn how to use the skin test anyway. So you might as well use it all the time. By 1911 Theodore Potter of Indiana University writes that “the eye reaction has already largely fallen into disuse, being replaced by the von Pirquet test.”
The eye test is still good for cattle, though!