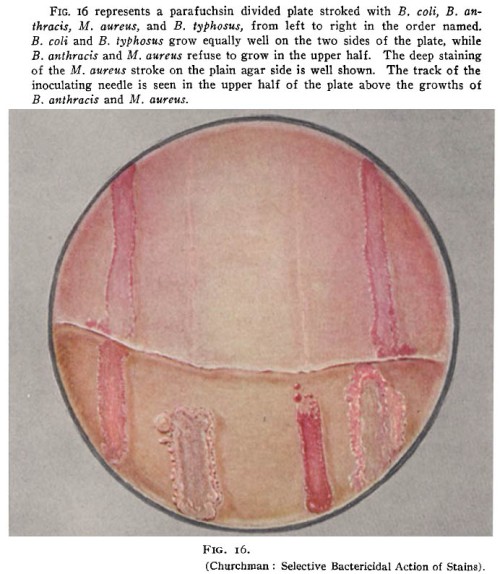
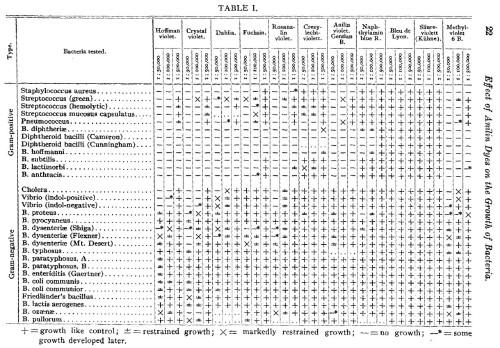
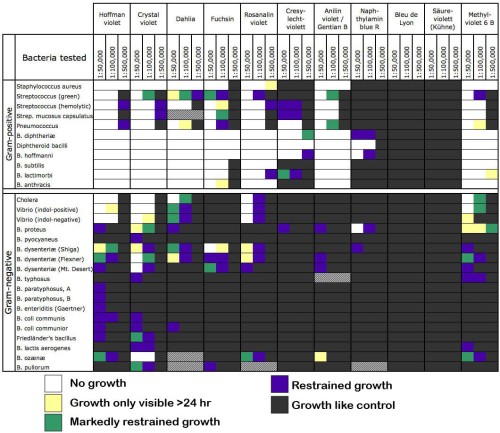
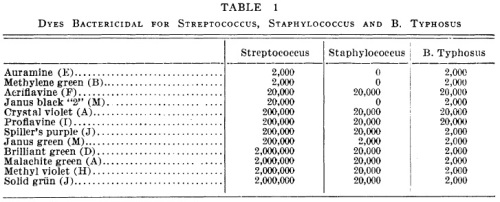
This week in bad ideas: Wart grafting
As described in this excerpt from James G. McCully’s Good Times In The Hospital, warts can be so extensive that they require a skin graft.
In the 19th century, skin grafts took many forms. By 1912 the process was down to a science, as detailed in Leonard Freeman’s Skin Grafting for Surgeons and General Practitioners. The basic method was established, with individual variations (“Donnelly, for some unexplained reason, prefers grafts from a portion of the skin subject to slight motion, such as the insertion of the deltoid muscle”), and complications and shortcomings were well known. (“Of all constitutional disorders, syphilis is probably the most disturbing, so much so that it has been claimed that grafting should never be attempted when this disease is present, and Freeman cites several cases which he thinks show that grafts will not adhere until syphilis has disappeared from the system.”) Many advancements had recently been made, even surprising doctors who didn’t quite expect them to work, and didn’t have a real theory to explain their success. The words of the late Dr. David Page (quoted here in 1872) were recalled with pity.
Freeman’s book, like other contemporaneous reviews of skin grafting techniques, includes an extensive section on “anomalies in skin grafting”. This included inducing blisters and transplanting the thin blister roof; transplanting hairs (taking care to include the hair sheath); skin from a wide range of different animals, particularly frogs; bits of muscle; egg membrane; and of course, thin sections of rabbit testes.
E. Aievoli made use of thin sections of the testes of rabbits for purposes of grafting in four cases, assuming that the testicle possesses a greater cellular activity than other portions of the body. The results were undoubtedly good, but it does not follow that they were better than could otherwise have been obtained.
It’s obvious now that most of these substances would not fuse with, or become, actual skin, but they provided a suitable covering for the wound / ulcer / burn until it could heal. From a 1909 review in the Boston Medical & Surgical Journal, by Dr. Albert Ehrenfried:
Orcel (1888) states his belief that animal grafts act merely as a sort of protective dressing, and he is substantiated by the experiments of Beresowsky (1892).
Ehrenfried has a particular interest in converting surgeons to the “Reverdin method”, which sounds simple, safe, and advantageous in that the source of the graft is easy to obtain. You just take “morsels” of existing skin, aiming for a maximum of 1/8 inch in diameter (but cut from as deep as possible), and “seed” them over the “granulations of ulcers”. Held in place with “diachylon plaster”, they should allow healing to proceed faster, as it will spread from these new pieces of skin as well as from the margins of the ulcer.
Reverdin preferred this to the even easier “epithelial dust” method of Mangoldt.
And another procedure, that might make more sense than the early 20th century writers think, is sponge grafting, “introduced by Hamilton in 1881.”
Sponge does not grow fast to the surface as does skin, but acts merely as a stimulating support for the granulations, finally undergoing complete absorption. The procedure is much inferior in its results to the transplantation of cuticle, and is now seldom employed.
A fine Turkey sponge is selected, soaked in dilute nitrohydrochloric acid until all calcareous particles have been dissolved, and then placed for a time in desired in a solution of potassium hydroxid. Very thin slices, which are the most serviceable, can be cut … and sterilized by boiling, or in a 5 per cent solution of carbolic acid.
The sponge is then spread upon the granulations, which have been rendered as nearly aseptic as possible (see the method of Reverdin), and dressed much as if it were a transplantation of skin. The granulations soon acquire new energy and push their way into the interstices of the sponge, which often almost disappears beneath them, so luxuriant is the growth.
Providing an aseptic framework for tissue to grow around is something bioengineers do now, right? Albeit not with skin tissue.
* * *
But there’s one skin-grafting method that might have extra negative effects, beyond introducing infection, irritation, or just wasting everyone’s time. At the top of this piece, we see how skin grafts may be necessary for a bad case of warts. But what if you have a bad case of burns, from molten iron spilling into your boot? You also need a skin graft… and as a source of the new skin, you could use warts!
The first (and only?) example of this procedure was described by Dr. Charles Leale of New York in an 1878 report in The Medical Record. Presumably this was the same Charles Leale who 12 years earlier had attended at the Lincoln assassination, as a 23-year-old army doctor who would soon leave the public eye for many decades of lucrative obscurity. (Obscure enough that in some of the journals that reprinted his observations on wart grafting, he was credited as Charles Seale or Charles Lale.)
The Medical Record seems not to be archived in Google Books. The most comprehensive summary of Leale’s “The Use of Common Warts of the Hand in Skin-Grafting” now available is probably in the September 1878 Atlanta Medical and Surgical Journal, vol. XVI(6):354-355. Here’s an abstract, and then the whole thing. Why not, it’s only two pages.
As common warts of the skin are collections of vascular papillae, admitting of easy separation without injury to their excessively thick layer of well-nourished epidermis, the idea was conceived that, by their use for the purpose of skin-grafting, better and more rapid results would be obtained than when the ordinary skin of less vitality is used. As proof of the theory, the following case is cited, where there had complete destruction of all the skin on the dorsum of the foot, involving to a great extent the deep cellular tissue, and where for several weeks no healing advanced until grafts of freshly removed warts from the patient’s hand immediately started little islands of tissue, which rapidly increased until they coalesced and met the margins of the border skin, thereby completely covering the foot by firm, protecting integument.
Do we know if it’s possible to transplant warts and have them take root in new places? If it didn’t happen in this case, as Leale says it didn’t, it may be very hard to do.